- Researchers blend historic physics with modern materials engineering to unlock durable, low-cost energy storage
- Study by Indian Institute of Technology Gandhinagar, Indian Institute of Science Education and Research Bhopal and global partners revives the classic Leidenfrost Effect for battery manufacturing
- Indium-tuned cathode architecture enables faster sodium-ion movement and exceptional durability
- Innovation could strengthen grid-scale renewable storage and support India’s clean-energy ambitions
NE SCIENCE & TECHNOLOGY BUREAU
GANDHINAGAR, MAR 6
A centuries-old physics curiosity—once observed in boiling kitchen pans—may soon power the next generation of affordable energy storage. Scientists from Indian Institute of Science Education and Research Bhopal, Indian Institute of Technology Gandhinagar, Swansea University, and the University of Southern Queensland have demonstrated how the Leidenfrost Effect, first described nearly 270 years ago by Johann Gottlob Leidenfrost, can be repurposed to create high-performance sodium-ion batteries for clean energy systems.
Their findings, recently published in the journal Small, illustrate how historical scientific insights can unlock modern solutions to one of the biggest challenges of the renewable energy era: affordable and durable battery storage.
Old Physics, New Energy Frontier
In the 18th century, Leidenfrost noticed that droplets of water placed on extremely hot metal surfaces skated around instead of instantly evaporating. The liquid formed a thin vapour cushion beneath it, allowing the droplet to glide across the surface.
This curious phenomenon—the Leidenfrost effect—has now found an unlikely application in advanced battery manufacturing.
“Lithium-ion batteries currently power everything from our smartphones to electric cars,” explained Rohit Ranganathan Gaddam, senior author of the study and Assistant Professor at IISER Bhopal.
“However, lithium is relatively rare and expensive to extract, making a greener, cost-effective alternative necessary.”
Why Sodium Could Power the Future
Researchers worldwide have been exploring sodium-ion batteries as a promising replacement for lithium technology. Sodium is abundant, inexpensive and widely available, making it attractive for large-scale renewable energy storage.
However, sodium ions are larger and heavier than lithium ions, which often causes them to strain and degrade battery materials during repeated charge cycles.
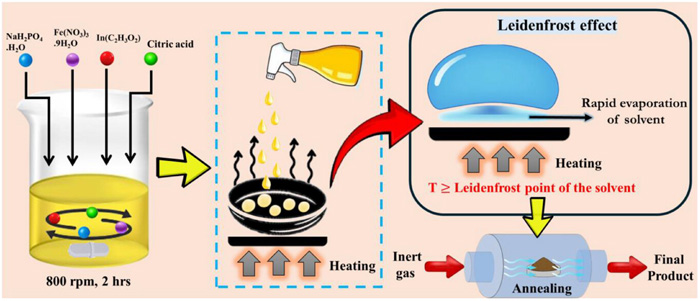
To overcome this, the research team engineered a specialised cathode material—Na₄Fe₃(PO₄)₂(P₂O₇)—known for its natural tunnel-like atomic structure.
“We decided to build the right cathode infrastructure—an atomic highway so sodium ions could zip through,” said Subhajit Singha, first author and PhD scholar at IISER Bhopal.
By replacing just one percent of iron atoms with indium, the researchers subtly expanded atomic spacing within the cathode. This modification allowed sodium ions to travel more efficiently while enhancing electrical conductivity.
Manufacturing Inspired by Leidenfrost Physics
The real innovation came during the material synthesis stage, where the team used the Leidenfrost effect itself.
“We tapped into the basics of the Leidenfrost effect to build cathode materials that outlast and outpace the standards currently in the market,” said Gaddam.
The researchers sprayed chemical droplets onto a superheated metal surface. Instead of sticking or burning, the droplets floated briefly on a vapour layer before rapidly evaporating and forming porous powder particles.
This fast and energy-efficient method eliminates the need for traditional high-temperature furnaces and produces sponge-like structures that absorb electrolytes more effectively—improving ion movement inside the battery.
Exceptional Performance and Durability
The new cathode material demonstrated energy density of around 359 Wh/kg and maintained stable performance for over 10,000 charge-discharge cycles.
“The optimised cathode material demonstrated remarkable durability,” said Raghavan Ranganathan, Associate Professor in the Department of Materials Engineering at Indian Institute of Technology Gandhinagar.
“Most consumer electronics batteries last only a few hundred cycles. Such longevity makes this technology ideal for renewable energy storage systems.”
Powering the Renewable Future
As India targets 500 GW of renewable energy capacity by 2030, affordable grid-scale battery storage is becoming critical for balancing solar and wind power supply.
The researchers believe their sodium-ion battery design could provide cost-effective storage for large energy networks, reducing dependence on lithium and strengthening sustainable supply chains.
“Our study shows that strategic atomic-level modification combined with a simple, scalable synthesis route can unlock performance previously out of reach for sodium-ion batteries,” said Gaddam.
Ranganathan added:
“This fusion of experimental and computational expertise across institutions and continents proves that historic scientific quirks like Leidenfrost can spark modern hubs of sustainable innovation.”
The study also aligns with global sustainability goals, including clean energy expansion and climate action, while offering a pathway toward affordable large-scale battery technologies.